External Control Arm for Relapsed/Refractory Multiple Myeloma
Patients diagnosed with multiple myeloma almost always relapse after treatment. Moreover, the time spent in remission is reduced after every relapse, indicating a constantly worsening condition of the patients . There are several available treatment regimens available for these refractory patients, and new agents and combinations of available therapies are being tested in clinical trials to identify optimal treatment pathways. An External Control Arm (ECA) may accelerate the understanding of which treatment is favourable to a selected patient group and provide an immediate clinical benefit.
The current case study shows how real-world data can be used to support a clinical trial through adoption of the target trial emulation framework. Specifically, we consider the ELOQUENT-2 study, an historic, randomised Phase III trial focusing on Elotuzumab Therapy for Relapsed or Refractory Multiple Myeloma (https://clinicaltrials.gov/ct2/show/NCT01239797). The aim of the case study is to develop an external control arm using RWD and compare it to the control arm derived in the trial and compare the analyses.
Methods
Arcturis operate on a unique model of entering into Strategic Research Agreements (SRAs) with NHS trusts across the UK to access de-identified patient records. For the current case study, data on UK NHS patients with a diagnosis of Multiple Myeloma (MM) were requested, including information on hospital activity, laboratory tests, systemic anti-cancer therapies (SACT), comorbidities, and key demographic characteristics, namely age, sex, and ethnicity.
Access to granular and longitudinal data allowed for a target trial emulation approach, which ensured that the study cohort matched to the studies inclusion and exclusion criteria, to ensure the clinical characteristics of the patients identified were similar to those recruited into the trial and looked to define observation periods similar to those in the trial setting. In addition, access to longitudinal laboratory data enabled the trial endpoint of progression-free survival (as defined by the International Myeloma Working Group (IMWG) criteria) to be replicated exactly as it would have been in the trial. Longitudinal changes in serum M protein, urine M protein, and kappa/lambda free-light chains, allowed for both relative and absolute changes in these markers over the course of the observational period to define the presence and date of disease progression. Descriptive statistics on the external control arm were derived and Kaplan-Meier estimators were used to determine time from initiation of treatment to progression or death. A Cox proportional hazards model was then used to compare the hazard function of the external control arm with the trials interventional arm.
Results
Arcturis retrieved data on 1,214 patients with an MM diagnosis. Out of the total MM sample, 208 were identified as having received lenalidomide and dexamethasone as a combination therapy. Of these, 83 were identified as meeting the selection criteria. The descriptive statistics comparing the trial control arm with the external control arm are provided in Table 1. Relative to the trial control arm, patients included in the external control arm had statistically similar proportions of females, patients of white ethnicity, and median number of previous therapies. However, patients in the external control arm were older, had a shorter disease duration and were more likely to score 1 or 2 for their functional status (WHO performance).
Table 1. The characteristics of patients in RWD, compared to the patients in the trial control arm.
| Trial Control Arm (n = 325) |
Synthetic Control Arm (n = 83) |
|
| Median age [range] | 66 [38, 91] | 72 [41, 93] |
| Female (%) | 41 | 43 |
| Ethnicity: White (%) | 86 | 82 |
| Number of prior therapy (median [range]) | 2 [1, 4] | 2 [1, 3] |
| Years since diagnosis (median [range]) | 3.5 [0.1, 16.2] | 1.4 [0.1, 6.3] |
| WHO performance (%) – 0 – 1 or 2 |
44 56 |
27 73 |
Progression-Free Survival
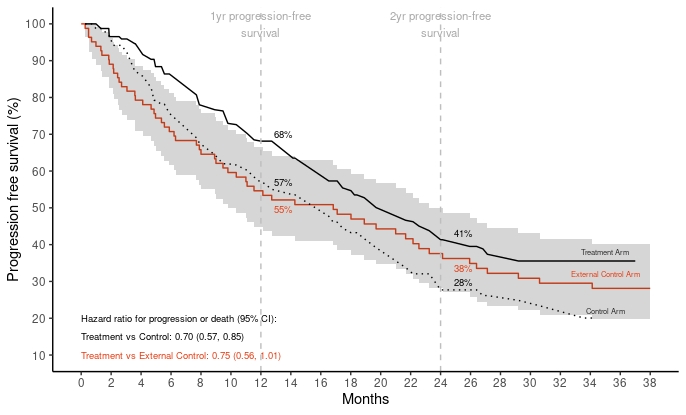
The cumulative incidence of PFS at 12 and 24-months in the external control arm was 55% (95% Confidence Interval (CI) 45-66) and 38% (95% CI: 28-49) respectively. In the trial arm, the PFS was slightly higher at 12-months compared to the ECA at 57% (95% CI: 51-62) and lower at 24-months at 27% (95% CI: 22-33). The cumulative incidence of the external control arm over the 38-month follow-up is depicted in Figure 1.
Cox regression analysis indicated a hazard ratio (HR) of 0.70 (95% CI 0.57 to 0.85; P<0.001) in the trial. Comparing the treatment arm with the external control arm indicated a similar HR of 0.75 (95% CI 0.56 to 1.01; P = 0.056).
Figure 1. Comparing hazards of progression or death between the trial and external controls.
Discussion
This case study shows how adoption of a target trial emulation approach using RWD can be used to imitate the control arm of a historical trial investigating the treatment efficacy of a new therapy regimen in the treatment of MM.
Whilst the hazard function and treatment effect produced similar estimates, the imbalances in the trial arms relative to the RWD and lower patient numbers affect the precision of the resulting treatment effect estimates. It is noteworthy that the current study did not have access to the individual patient level data of the trial. However, having such access would allow introducing further precision into analyses; either by selecting ECA patients in a stringent matching or allow the use of advanced statistical techniques to adjust for the differences between an ECA and an experimental arm, namely Inverse Probability Treated Weights (IPTW) and other propensity matching methods.
In conclusion, ECAs are likely to further grow in utilisation and value in bringing new treatments to patients, including digital therapeutics. They are especially an attractive option when aiming to reduce the cost and length of a clinical trial. We see developing use cases similar to ours as key to identify therapeutic areas that are most suited for the use of ECAs, as well as developing methodological expertise in combining RWD and clinical trial data under the target trial emulation framework to support further advancement and use of ECAs as part of clinical development of new therapeutics.